12.03.21
4100 Frontage Rd S
Lakeland, FL 33815
863-688-1200
inquiries@ACIN95.com
www.acipak.com / www.acin95.com
In March 2020, a Florida-based manufacturing company stepped up to meet the needs of a local hospital group to help in the fight against Covid-19. At that time, Lakeland-based Advanced Concept Innovations (ACI) was contacted by Lakeland Regional Hospital with an emergency request for personal protective equipment (PPE) of all types. With capabilities as a medical device packager and manufacturer since 2004, ACI’s engineering and manufacturing teams were quickly able to respond with a face shield design, which it was able to start producing within a week of the request.
ACI was also asked if it could fill the desperate need for face masks. As a nonwoven converter since 2005, ACI had expertise in the conversion and assembly of nonwoven materials into consumer products, but did not have equipment designed for face mask manufacturing.
“Our engineering team reviewed the different styles of face masks, and determined within 24 hours that we could redesign, retool and rebuild several of our converting lines into a new ultra-high-speed line, to be able to manufacture a duckbill style N95 respirator,” says Matt Muller, president of ACI.
ACI’s internal teams, along with its external vendors, immediately started disassembling the existing equipment and designing, manufacturing and programming a high speed N95 respirator line. “We started producing general purpose respirators in May 2020, 45 days after beginning the equipment redesign, with NIOSH approved N95 Respirator production following up in June, approximately 60 days after the start of the redesign,” Muller says.
ACI’s respirators are a 10-layer design, comprised of four layers of meltblown, four layers of spunbond and two elastomeric headband layers. Today, its capacity is in excess of 70 million N95 surgical respirators per month—840 million annually.
According to Muller, the current market is saturated with non-medical, non-domestic N95 respirators, as these have been permitted for use by the existing EUA (Emergency Use Authorization) issued by the FDA (the EUA was recently revoked for KN95 respirators). “Hospitals are also permitting single use respirators to be used for extended periods, outside of their intended design and use,” Muller continues. “These non-medical respirators are putting our healthcare and first responders at risk, as they are not configured for healthcare settings. Once the EUA is lifted by the FDA, and the healthcare systems begin proper use of the respirators, the market will normalize for single-use surgical N95s.”
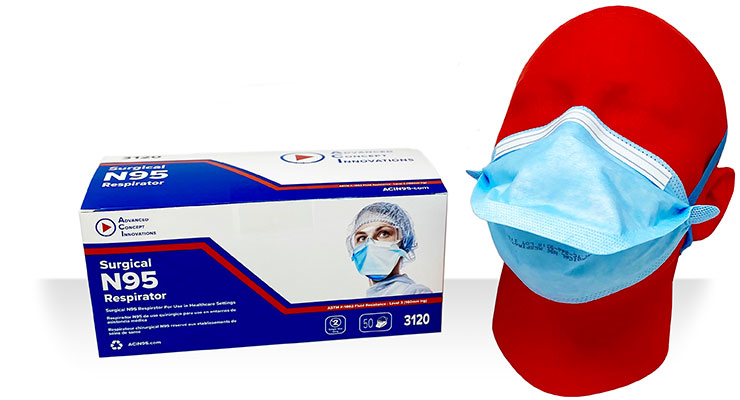
ACI’s 3120 Single Use Surgical N95 Respirator is produced in Lakeland, FL, where its 315,000 square foot facility has been registered with the FDA since 2006. Not only are ACI’s respirators NIOSH approved (TC-84A-9318), but they have also been cleared by the FDA which makes them approved for use in healthcare settings, even after the EUA is lifted. ACI’s Surgical N95 conforms to recognized standards for flammability, fluid resistance and biocompatibility. Its fluid resistance is a Level 3, which is the highest level of protection (160 mm Hg).
“Domestic supply and manufacturing of surgical N95 respirators provide a secure supply chain for our healthcare workers and first responders,” says Kevin Pulcini, senior vice president, Business Development. “We cannot rely on other countries, whose self-interest is contrary to ours, to hold us hostage during emergency situations. Even when foreign product is available, supply chain logistics are extremely cumbersome and time consuming.”
ACI believes the pandemic has forever changed the manufacturing supply chain in the U.S., however, there is a cautionary tale, Muller says. “Although domestic manufacturing was spun up by the major manufacturers at the beginning of the pandemic, as soon as the market became somewhat saturated, they retreated back to their offshore manufacturing, further reducing their new domestic capability. We must produce domestically manufactured PPE at competitive costs to ensure we maintain sufficient capacity domestically.”
ACI has been able to meet this competitive need by redesigning the traditional production methods used for manufacturing N95 respirators. “We need to continue using technology and innovation to redefine the labor-intensive Asian production model for PPE, to enable further domestic capabilities, without government subsidies,” Pulcini adds.
Lakeland, FL 33815
863-688-1200
inquiries@ACIN95.com
www.acipak.com / www.acin95.com
In March 2020, a Florida-based manufacturing company stepped up to meet the needs of a local hospital group to help in the fight against Covid-19. At that time, Lakeland-based Advanced Concept Innovations (ACI) was contacted by Lakeland Regional Hospital with an emergency request for personal protective equipment (PPE) of all types. With capabilities as a medical device packager and manufacturer since 2004, ACI’s engineering and manufacturing teams were quickly able to respond with a face shield design, which it was able to start producing within a week of the request.
ACI was also asked if it could fill the desperate need for face masks. As a nonwoven converter since 2005, ACI had expertise in the conversion and assembly of nonwoven materials into consumer products, but did not have equipment designed for face mask manufacturing.
“Our engineering team reviewed the different styles of face masks, and determined within 24 hours that we could redesign, retool and rebuild several of our converting lines into a new ultra-high-speed line, to be able to manufacture a duckbill style N95 respirator,” says Matt Muller, president of ACI.
ACI’s internal teams, along with its external vendors, immediately started disassembling the existing equipment and designing, manufacturing and programming a high speed N95 respirator line. “We started producing general purpose respirators in May 2020, 45 days after beginning the equipment redesign, with NIOSH approved N95 Respirator production following up in June, approximately 60 days after the start of the redesign,” Muller says.
ACI’s respirators are a 10-layer design, comprised of four layers of meltblown, four layers of spunbond and two elastomeric headband layers. Today, its capacity is in excess of 70 million N95 surgical respirators per month—840 million annually.
According to Muller, the current market is saturated with non-medical, non-domestic N95 respirators, as these have been permitted for use by the existing EUA (Emergency Use Authorization) issued by the FDA (the EUA was recently revoked for KN95 respirators). “Hospitals are also permitting single use respirators to be used for extended periods, outside of their intended design and use,” Muller continues. “These non-medical respirators are putting our healthcare and first responders at risk, as they are not configured for healthcare settings. Once the EUA is lifted by the FDA, and the healthcare systems begin proper use of the respirators, the market will normalize for single-use surgical N95s.”
ACI’s 3120 Single Use Surgical N95 Respirator is produced in Lakeland, FL, where its 315,000 square foot facility has been registered with the FDA since 2006. Not only are ACI’s respirators NIOSH approved (TC-84A-9318), but they have also been cleared by the FDA which makes them approved for use in healthcare settings, even after the EUA is lifted. ACI’s Surgical N95 conforms to recognized standards for flammability, fluid resistance and biocompatibility. Its fluid resistance is a Level 3, which is the highest level of protection (160 mm Hg).
“Domestic supply and manufacturing of surgical N95 respirators provide a secure supply chain for our healthcare workers and first responders,” says Kevin Pulcini, senior vice president, Business Development. “We cannot rely on other countries, whose self-interest is contrary to ours, to hold us hostage during emergency situations. Even when foreign product is available, supply chain logistics are extremely cumbersome and time consuming.”
ACI believes the pandemic has forever changed the manufacturing supply chain in the U.S., however, there is a cautionary tale, Muller says. “Although domestic manufacturing was spun up by the major manufacturers at the beginning of the pandemic, as soon as the market became somewhat saturated, they retreated back to their offshore manufacturing, further reducing their new domestic capability. We must produce domestically manufactured PPE at competitive costs to ensure we maintain sufficient capacity domestically.”
ACI has been able to meet this competitive need by redesigning the traditional production methods used for manufacturing N95 respirators. “We need to continue using technology and innovation to redefine the labor-intensive Asian production model for PPE, to enable further domestic capabilities, without government subsidies,” Pulcini adds.