Tara Olivo, Associate Editor07.31.17
The adoption of nonwovens in the medical market is on an upward swing. Used for surgical drapes and gowns, face masks, wound care dressings, wipes, absorbent hygiene products and other fields, the medical market is expected to grow at an exceptional rate considering the increased demand from the healthcare industry for these types of products, according to information from market researcher MarketsandMarkets.
“This market has taken a huge leap forward with the ability of nonwoven products demonstrating high resistance to contaminations, exhibiting absorbency and barrier properties and ensuring comfort while in use,” says Neha Sharma, research analyst at MarketsandMarkets in Packaging, Construction, and Mining. “The healthcare industry has significantly transited from reusable medical fabrics to disposable, nonwoven-based fabrics across the world. This high demand for medical nonwoven products can be credited to the increase in the adoption of infection control measures by the medical community.”
The shift in trend from reusable medical products to single-use nonwoven products is due to the fact that the microbial penetration resistance of single-use medical nonwoven products is very high compared to reusable ones, she points out.
In a study on the Medical Packaging Market, MarketsandMarkets explored the use of nonwovens in medical applications. Nonwovens—a major raw material in medical packaging—recorded a market size of $5.46 billion in 2016 and is expected to reach $7.23 billion by 2022. The researcher says this market is anticipated to grow at a CAGR of 4.8% between 2017 to 2022.
“Rapid advancements in the medical procedures and technologies have fostered the demand for nonwovens in the medical sector,” Sharma explains. “Medical fabrics are required to possess sufficient strength so that they can resist tearing and can repel body fluids, including blood. Nonwoven fabrics, owing to their high liquid resistance capabilities, tensile strength and hydrophobic/hydrophilic properties, have gained high traction in the medical industry. Medical nonwoven manufacturers are focusing on the introduction of new products with improved barrier protection.”
Marika Mäkilä, Suominen’s product manager, Medical, also discusses the many favorable qualities of single-use nonwoven based products over their reusable counterparts. Among them are performance and engineered properties, cleanliness, disposability of contaminated products and related safety aspects. “Depending on the end product, preferred and beneficial functionalities may be e.g. better softness, conformability, lower linting or liquid absorption,” she adds.
The Finnish nonwovens producer recently added Fibrella Wrap to its medical range of products in South America. A soft and skin-friendly material, Fibrella Wrap can be used for paddings used under casts to protect bones and for any wound care application where cushion is needed. It is a stretchy nonwoven material that conforms well to body contours, making the patient feel more comfortable when wearing a cast.
“In South America, cotton wadding is the most typical material used under casts and in wound care but it has two major weaknesses: it has no strength and it can be difficult to apply for the clinicians,” Mäkilä explains. “Fibrella Wrap is strong enough to be applied in a circular fashion while can also be easily torn when needed.”
Fibrella Wrap utilizes Suominen’s recent investments in upgraded production technology in its Paulínia, Brazil, plant.
Meanwhile, Freudenberg Performance Materials, which focuses on the wound care segment of the medical market, has been able to expand its technology base since it acquired polyurethane (PU) foam manufacturer Polymer Health Technologies in 2015.
Dr. Frank Heislitz, CTO Freudenberg Performance Materials, says Freudenberg is one of the first companies in the market to have developed an MDI-based PU foam. “The innovation significantly surpasses the performance of commonly used TDI-based PU foams,” he explains. “With the new MDI-based PU foam, customers are able to offer modern wound dressings for chronic venous leg ulcers, which absorb wound fluids much faster and have significantly better retention properties. In addition, these wound dressings are much stronger.”
TDI describes the chemistry behind the PU system and stands for Toluol-2,4-diisocyanat. Currently this is the standard chemical PU system in the market for PU foams in wound care and has been in use successfully for roughly the last 15 years, according to Freudenberg.
“However, recently wound care OEMs have been working on innovation in PU foam dressings that would offer enhanced performance compared to the TDI chemistry such as: Improvement in retention of fluid; stronger foam; soft and conformable; fast vertical wicking; and different swelling properties,” Heislitz says. “These characteristics can be met with PU foam dressings based on new MDI (Methylendiphenylisocyanate) chemistry.”
Growing in Medical
Texol, a manufacturer of specialty topsheet and elastic material for hygiene applications with sites in Italy and Chile, has purchased the majority of ORMA, a maker of hydroentanglement specialty products for the medical, tablecloth, cosmetics and wipes industry.
Texol says this purchase is part of its commitment to develop and produce innovative and specialty products for markets beyond hygiene and increasing its market presence in other areas.
Together, the two companies will produce and supply products taking and combining the advantages and synergies of Texol know-how, experience in technologies and product development, with ORMA’s expertise and capabilities in production, logistics and market knowledge.
Because of its financial strength, Texol says it will continue to make important investments, especially in bio and green products. Ecology and sustainability will be one of the main focuses for Texol’s and ORMA’s industrial green philosophy for future development. In collaboration with their suppliers and customers, they are looking for very low environmental impact products offering practical sustainable solutions in an increasingly demanding and selective market.
On the end-product side, Essity, formerly SCA’s hygiene business, is moving beyond manufacturing only adult incontinence products for the healthcare market. In the spring, the company closed its acquisition of BSN medical, a medical solutions company that develops, manufactures, markets and sells products within advanced and acute wound care, compression therapy and orthopedics. With this acquisition, Essity gained the healthcare brands Leukoplast, Cutimed, JOBST, Delta Cast, Delta Lite and Actimove.
BSN medical is now included in SCA’s Personal Care business area. Together, SCA’s Incontinence Care unit and BSN medical form the new business unit Health and Medical Solutions, according to Katarina Regårdh Bengtsson, communications director, Health and Medical Solutions, Essity.
“BSN medical’s product portfolio is complementary to Essity’s incontinence products offering in terms of product complexity and go to market in important channels within healthcare such as hospitals and pharmacies,” she explains. “This will further strengthen and develop our global market leading position within the incontinence segment under the Tena brand. At the same time, the BSN medical business will benefit from our strong presence in both healthcare and retail sales channels as well as our extensive knowledge in brand building and focus on digitalization.”
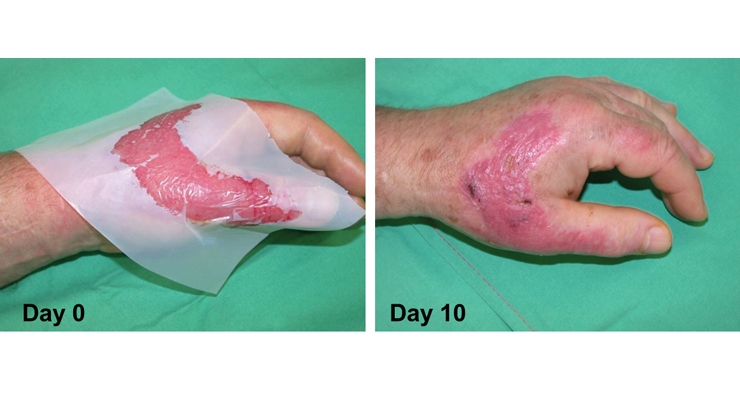
In order to increase the quality of life of affected patients with complex or hard-to-heal wounds, BSN medical offers product solutions that often feature nonwovens to support tissue growth, pain reduction and to manage different levels of exudate.
“In recent years, innovative manufacturing methods from mechanical to bioreactor based provided nonwovens with unique fiber combinations and structures. These nonwovens with manifold and adjustable characteristics offer outstanding solutions for modern wound healing, especially for complex wounds,” explains Dr.-Ing. Annahit Arshi, director of Biomaterials & Medical Devices, Global Research & Development, BSN medical.
For example, she says, nanofiber-nonwovens highly resemble human tissue, and an application of such a structure can provide a scaffold-like surface for cell ingrowth. BSN’s Cutimed Epiona, a nano-nonwoven collagen, absorbent wound dressing provides a structure similar to the human dermis that can be demonstrated with microscopic images.
“The collagen nonwoven is obtained from native bovine that has been passed through a cryogenic process,” Arshi says. “The resulting 3D structure enables cells to easily grow in and helps cell migration as well as formation of the body’s own collagen fibrils and fibers. Thereby, Cutimed Epiona is supporting the formation of granulation tissue.”
Meanwhile, for skin donor sites or burns, BSN has developed a nanofiber nonwoven called Cutimed Epigraft. In treating these types of wounds, it’s vital to temporarily replace the skin function, Arshi says. Cuticell Epigraft is able to maintain an antimicrobial barrier, while offering moisture vapor to pass through, thereby supporting the body’s fluid homeostasis. Additionally, the nanostructured surface is able to reduce pain and support the process of re-epithelization. “The dressing remains onto the wound until healing is completed, thereby superseding disturbing dressing changes,” she explains.
According to Arshi, bacterial nanocellulose – the basis for Cuticell Epigraft – that is extracellularly synthesized by specific bacterial strains, organizes in randomly assembled tangled structures, having fiber dimensions of 20-100 nm width and 1.0-5.0 µm length. “The fibers are produced in a wet environment and create a gel with a content of approximately 1% highly pure cellulose,” she says. “The resulting nonwoven combines the high tear strength of cellulose with a large surface area, which enables cells to directly interact with the dressing. In a dried state bacterial nanocellulose appears like simple thin paper, but its specific nanostructure can cover open nerve endings in the wound for rapid pain relief and moreover creates an ideal environment for granulation and re-epithelialization.”
Reducing Waste
Halyard Health, a producer of surgical gowns, drapes, sterile wraps and apparel, announced in May that it formed a license and supply agreement with Sustainable Solutions LLC to convert recycled Halyard Wrap, made of SMS nonwoven fabric, into environmentally conscious products for healthcare facilities.
This agreement is an extension of Halyard’s Blue Renew Wrap Recycling Program, designed to offer healthcare facilities a step-by-step approach for easily and efficiently recycling the Halyard’s used sterilization wrap. Sustainable Solutions will collect and recycle Halyard Wrap waste from customers and convert the material into BlueCON Resin—the company’s proprietary material engineered from clean Halyard Wrap waste. Ultimately, Sustainable Solutions will convert the resin into sustainable hospital products, including distribution and product totes, garbage cans, bedpans and washbasins.
“This new effort with Sustainable Solutions underscores our collective commitment to develop innovative solutions that enable healthcare facilities to take concrete steps to reduce their carbon footprint and achieve their overarching sustainability goals,” says Joe Hannibal, associate marketing director, Halyard Health. “Best of all, it represents a natural extension of our Blue Renew Wrap Recycling Program.”
In Halyard’s overall Blue Renew recycling program, there are currently more than 400 hospitals in the U.S. recycling Halyard Wrap, with more than three million pounds being recycled.
“This market has taken a huge leap forward with the ability of nonwoven products demonstrating high resistance to contaminations, exhibiting absorbency and barrier properties and ensuring comfort while in use,” says Neha Sharma, research analyst at MarketsandMarkets in Packaging, Construction, and Mining. “The healthcare industry has significantly transited from reusable medical fabrics to disposable, nonwoven-based fabrics across the world. This high demand for medical nonwoven products can be credited to the increase in the adoption of infection control measures by the medical community.”
The shift in trend from reusable medical products to single-use nonwoven products is due to the fact that the microbial penetration resistance of single-use medical nonwoven products is very high compared to reusable ones, she points out.
In a study on the Medical Packaging Market, MarketsandMarkets explored the use of nonwovens in medical applications. Nonwovens—a major raw material in medical packaging—recorded a market size of $5.46 billion in 2016 and is expected to reach $7.23 billion by 2022. The researcher says this market is anticipated to grow at a CAGR of 4.8% between 2017 to 2022.
“Rapid advancements in the medical procedures and technologies have fostered the demand for nonwovens in the medical sector,” Sharma explains. “Medical fabrics are required to possess sufficient strength so that they can resist tearing and can repel body fluids, including blood. Nonwoven fabrics, owing to their high liquid resistance capabilities, tensile strength and hydrophobic/hydrophilic properties, have gained high traction in the medical industry. Medical nonwoven manufacturers are focusing on the introduction of new products with improved barrier protection.”
Marika Mäkilä, Suominen’s product manager, Medical, also discusses the many favorable qualities of single-use nonwoven based products over their reusable counterparts. Among them are performance and engineered properties, cleanliness, disposability of contaminated products and related safety aspects. “Depending on the end product, preferred and beneficial functionalities may be e.g. better softness, conformability, lower linting or liquid absorption,” she adds.
The Finnish nonwovens producer recently added Fibrella Wrap to its medical range of products in South America. A soft and skin-friendly material, Fibrella Wrap can be used for paddings used under casts to protect bones and for any wound care application where cushion is needed. It is a stretchy nonwoven material that conforms well to body contours, making the patient feel more comfortable when wearing a cast.
“In South America, cotton wadding is the most typical material used under casts and in wound care but it has two major weaknesses: it has no strength and it can be difficult to apply for the clinicians,” Mäkilä explains. “Fibrella Wrap is strong enough to be applied in a circular fashion while can also be easily torn when needed.”
Fibrella Wrap utilizes Suominen’s recent investments in upgraded production technology in its Paulínia, Brazil, plant.
Meanwhile, Freudenberg Performance Materials, which focuses on the wound care segment of the medical market, has been able to expand its technology base since it acquired polyurethane (PU) foam manufacturer Polymer Health Technologies in 2015.
Dr. Frank Heislitz, CTO Freudenberg Performance Materials, says Freudenberg is one of the first companies in the market to have developed an MDI-based PU foam. “The innovation significantly surpasses the performance of commonly used TDI-based PU foams,” he explains. “With the new MDI-based PU foam, customers are able to offer modern wound dressings for chronic venous leg ulcers, which absorb wound fluids much faster and have significantly better retention properties. In addition, these wound dressings are much stronger.”
TDI describes the chemistry behind the PU system and stands for Toluol-2,4-diisocyanat. Currently this is the standard chemical PU system in the market for PU foams in wound care and has been in use successfully for roughly the last 15 years, according to Freudenberg.
“However, recently wound care OEMs have been working on innovation in PU foam dressings that would offer enhanced performance compared to the TDI chemistry such as: Improvement in retention of fluid; stronger foam; soft and conformable; fast vertical wicking; and different swelling properties,” Heislitz says. “These characteristics can be met with PU foam dressings based on new MDI (Methylendiphenylisocyanate) chemistry.”
Growing in Medical
Texol, a manufacturer of specialty topsheet and elastic material for hygiene applications with sites in Italy and Chile, has purchased the majority of ORMA, a maker of hydroentanglement specialty products for the medical, tablecloth, cosmetics and wipes industry.
Texol says this purchase is part of its commitment to develop and produce innovative and specialty products for markets beyond hygiene and increasing its market presence in other areas.
Together, the two companies will produce and supply products taking and combining the advantages and synergies of Texol know-how, experience in technologies and product development, with ORMA’s expertise and capabilities in production, logistics and market knowledge.
Because of its financial strength, Texol says it will continue to make important investments, especially in bio and green products. Ecology and sustainability will be one of the main focuses for Texol’s and ORMA’s industrial green philosophy for future development. In collaboration with their suppliers and customers, they are looking for very low environmental impact products offering practical sustainable solutions in an increasingly demanding and selective market.
On the end-product side, Essity, formerly SCA’s hygiene business, is moving beyond manufacturing only adult incontinence products for the healthcare market. In the spring, the company closed its acquisition of BSN medical, a medical solutions company that develops, manufactures, markets and sells products within advanced and acute wound care, compression therapy and orthopedics. With this acquisition, Essity gained the healthcare brands Leukoplast, Cutimed, JOBST, Delta Cast, Delta Lite and Actimove.
BSN medical is now included in SCA’s Personal Care business area. Together, SCA’s Incontinence Care unit and BSN medical form the new business unit Health and Medical Solutions, according to Katarina Regårdh Bengtsson, communications director, Health and Medical Solutions, Essity.
“BSN medical’s product portfolio is complementary to Essity’s incontinence products offering in terms of product complexity and go to market in important channels within healthcare such as hospitals and pharmacies,” she explains. “This will further strengthen and develop our global market leading position within the incontinence segment under the Tena brand. At the same time, the BSN medical business will benefit from our strong presence in both healthcare and retail sales channels as well as our extensive knowledge in brand building and focus on digitalization.”
In order to increase the quality of life of affected patients with complex or hard-to-heal wounds, BSN medical offers product solutions that often feature nonwovens to support tissue growth, pain reduction and to manage different levels of exudate.
“In recent years, innovative manufacturing methods from mechanical to bioreactor based provided nonwovens with unique fiber combinations and structures. These nonwovens with manifold and adjustable characteristics offer outstanding solutions for modern wound healing, especially for complex wounds,” explains Dr.-Ing. Annahit Arshi, director of Biomaterials & Medical Devices, Global Research & Development, BSN medical.
For example, she says, nanofiber-nonwovens highly resemble human tissue, and an application of such a structure can provide a scaffold-like surface for cell ingrowth. BSN’s Cutimed Epiona, a nano-nonwoven collagen, absorbent wound dressing provides a structure similar to the human dermis that can be demonstrated with microscopic images.
“The collagen nonwoven is obtained from native bovine that has been passed through a cryogenic process,” Arshi says. “The resulting 3D structure enables cells to easily grow in and helps cell migration as well as formation of the body’s own collagen fibrils and fibers. Thereby, Cutimed Epiona is supporting the formation of granulation tissue.”
Meanwhile, for skin donor sites or burns, BSN has developed a nanofiber nonwoven called Cutimed Epigraft. In treating these types of wounds, it’s vital to temporarily replace the skin function, Arshi says. Cuticell Epigraft is able to maintain an antimicrobial barrier, while offering moisture vapor to pass through, thereby supporting the body’s fluid homeostasis. Additionally, the nanostructured surface is able to reduce pain and support the process of re-epithelization. “The dressing remains onto the wound until healing is completed, thereby superseding disturbing dressing changes,” she explains.
According to Arshi, bacterial nanocellulose – the basis for Cuticell Epigraft – that is extracellularly synthesized by specific bacterial strains, organizes in randomly assembled tangled structures, having fiber dimensions of 20-100 nm width and 1.0-5.0 µm length. “The fibers are produced in a wet environment and create a gel with a content of approximately 1% highly pure cellulose,” she says. “The resulting nonwoven combines the high tear strength of cellulose with a large surface area, which enables cells to directly interact with the dressing. In a dried state bacterial nanocellulose appears like simple thin paper, but its specific nanostructure can cover open nerve endings in the wound for rapid pain relief and moreover creates an ideal environment for granulation and re-epithelialization.”
Reducing Waste
Halyard Health, a producer of surgical gowns, drapes, sterile wraps and apparel, announced in May that it formed a license and supply agreement with Sustainable Solutions LLC to convert recycled Halyard Wrap, made of SMS nonwoven fabric, into environmentally conscious products for healthcare facilities.
This agreement is an extension of Halyard’s Blue Renew Wrap Recycling Program, designed to offer healthcare facilities a step-by-step approach for easily and efficiently recycling the Halyard’s used sterilization wrap. Sustainable Solutions will collect and recycle Halyard Wrap waste from customers and convert the material into BlueCON Resin—the company’s proprietary material engineered from clean Halyard Wrap waste. Ultimately, Sustainable Solutions will convert the resin into sustainable hospital products, including distribution and product totes, garbage cans, bedpans and washbasins.
“This new effort with Sustainable Solutions underscores our collective commitment to develop innovative solutions that enable healthcare facilities to take concrete steps to reduce their carbon footprint and achieve their overarching sustainability goals,” says Joe Hannibal, associate marketing director, Halyard Health. “Best of all, it represents a natural extension of our Blue Renew Wrap Recycling Program.”
In Halyard’s overall Blue Renew recycling program, there are currently more than 400 hospitals in the U.S. recycling Halyard Wrap, with more than three million pounds being recycled.